It is also very important to obtain a result of zero upon adding the oxidation states of all atoms of a neutral molecule. A pure element will always have an oxidation state of zero. The oxidation state is as high as +8 in certain elements, while it is as low as -4. Reduction refers to a reaction where oxidation decreases. Oxidation and reduction are the two reactions that help in, formally transferring electrons. The term was developed by Antoine Lavoisier. The state can either be positive, zero, or negative. The state of oxidation of a specific element refers to the degree to which an atom can lose or gain its electrons. 
The oxidation state of elements in the periodic table The chemical reactivity on moving from top to bottom increases, and it decreases on moving across. The substance can react with itself or with some other substance and release energy. Hence their ability to conduct electricity is determined.Ĭhemical Reactivity – it refers to the energy or the force of a chemical substance to undergo a reaction. Metallic character – the metallic character of elements in a periodic table tends to increase on moving from top to bottom, and it tends to decrease on moving from the left towards the right. It increases on moving from left to right as more energy is released. The affinity of electrons – refers to the energy that is released when an electron is gained by an atom. On moving in a diagonal direction, atomic radius tends to increase. On moving from left to right, it increases, and on moving from top to bottom, it increases. Ionisation energy increases on moving horizontally across a period, whereas on moving in the downward direction, the ionisation energy decreases.Ītomic radii – the distance between the outermost electron and the nucleus is known as the atomic radius. Ionisation energy – this energy helps the atoms to remove the electrons. An exception in group 3 elements is present where electronegativity increases towards thallium. On moving in the downward direction, the electronegativity decreases. On moving from left to right horizontally in a periodic table, there is an increase in electronegativity. The Pauling scale is used to measure electronegativity. Some periodic trends and chemical reactivity in the periodic table are as follows –Įlectronegativity – measures the ability of a molecule to gain electrons within a chemical bond. The trends of the elements in a periodic table depend on various factors like electronegativity, ionisation energy, electron affinity, atomic radii, metallic character, and chemical reactivity. Periodic trends refer to the patterns of the properties of the chemical elements. The periodic trends and chemical reactivities of elements are shared below. The gas elements do not gain or lose their electrons because the electronic configuration is filled. Hence the elements on the left side in the downwards direction are the highly reactive elements. In a period, the chemical reactivity decreases on moving towards the right, whereas in a group, chemical reactivity increases on moving downwards. Hence the electronegativity and the ionisation energy determine a chemical reaction. The higher the reactivity of the element, the easier it is to combine. The electrons between the elements are shared, or the elements lose or gain the electrons. 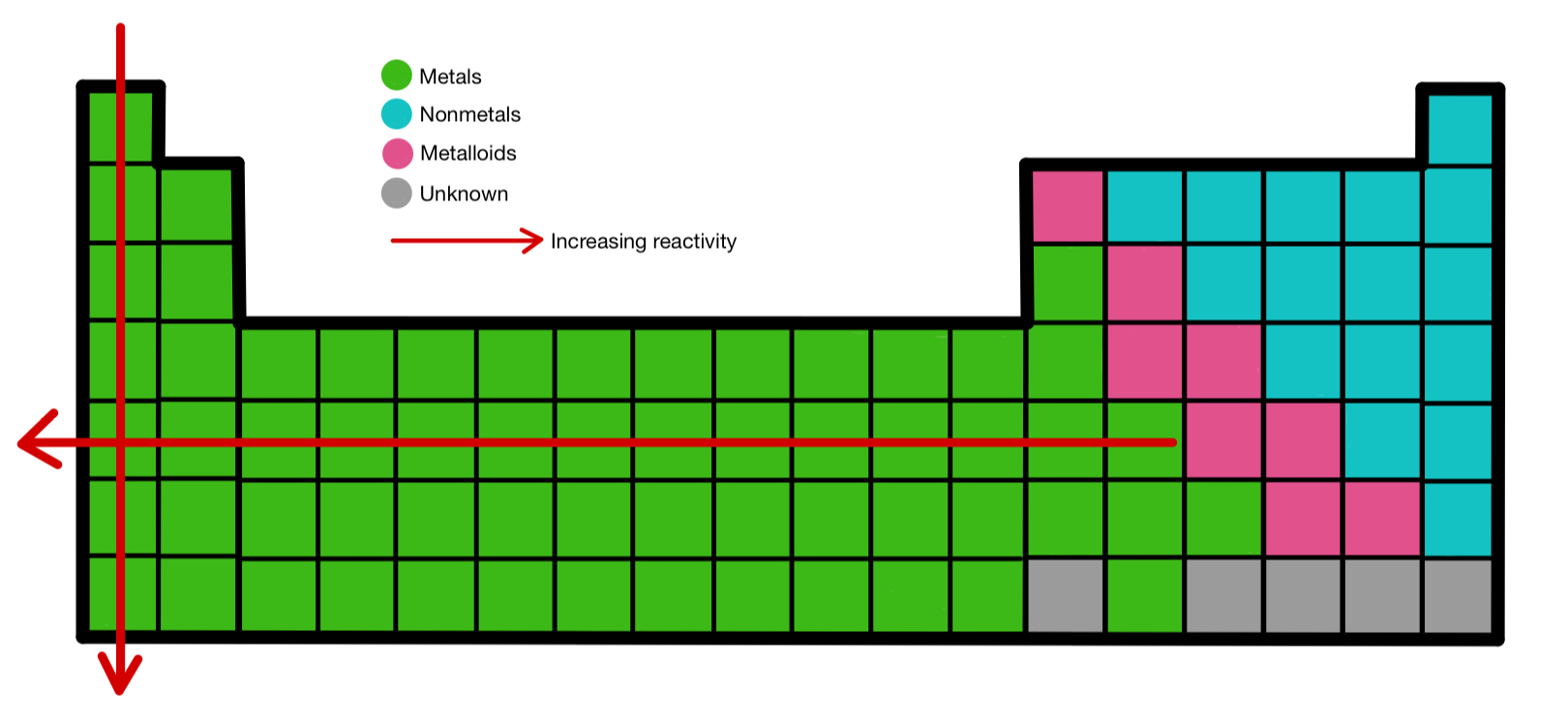
(4) Transition elements are known as metals they are less electropositive than group-1 and group-2.The rate at which a chemical substance or an element undergoes a chemical reaction is known as reactivity. C O, N O, N 2 O ).Īmphoteric oxides behave as acidic with bases and as basic with acids, whereas neutral oxides have no acidic or basic properties. Oxides of elements in the centre are amphoteric. The element on extreme right is the most acidic. The normal oxide formed by the element on extreme left is the most basic. (3) Reactivity with Oxygen : Elements on two extremes of a period easily combine with oxygen to from oxides. 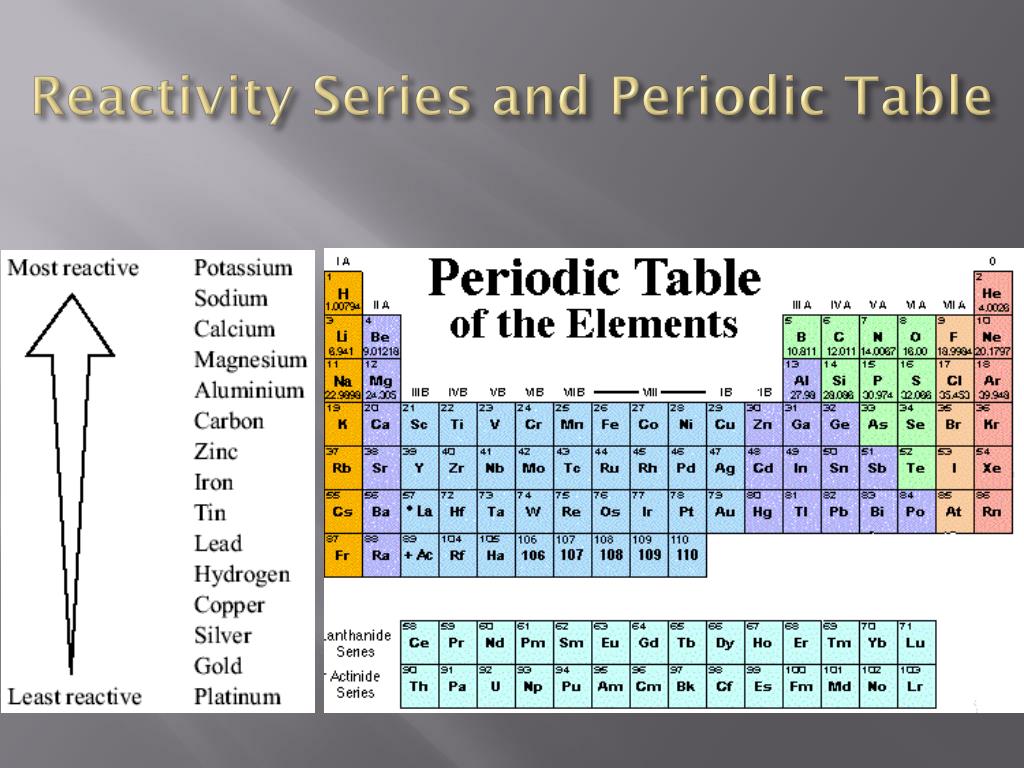
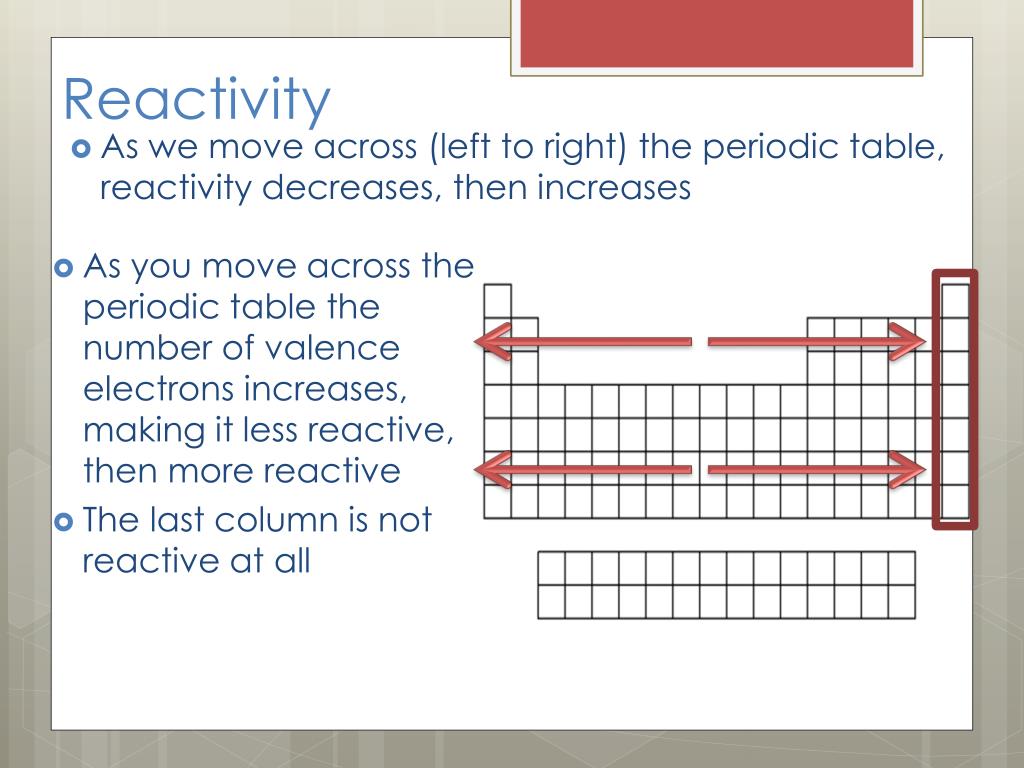
(2) Metallic and non-metallic Properties : We move left to right in a period metallic property decreases and non-metallic property increases. (1) Reactivity of Oxidation and Reduction : Extremely left elements of periodic table are reducing agent and extremely right elements of periodic table are oxidising agent. (Exception : Noble gas)Įlements are more reactive in both of the side and elements are less reactive in middle in periodic table.Įlements are situated very left side (Alkali metals) have tendency to lose one electron and elements are situated right side (Halogen) have tendency to gain one electron and becomes negative. So, we can say that in period ionisation enthalpy is very less in left side and electron gain enthalpy is very high and negative in right side. So, ionisation enthalpy increases in period and value of electron gain enthalpy becomes more negative. When we go left to right atomic radius and ionic radius decreases. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
